FDA Grants Breakthrough Device
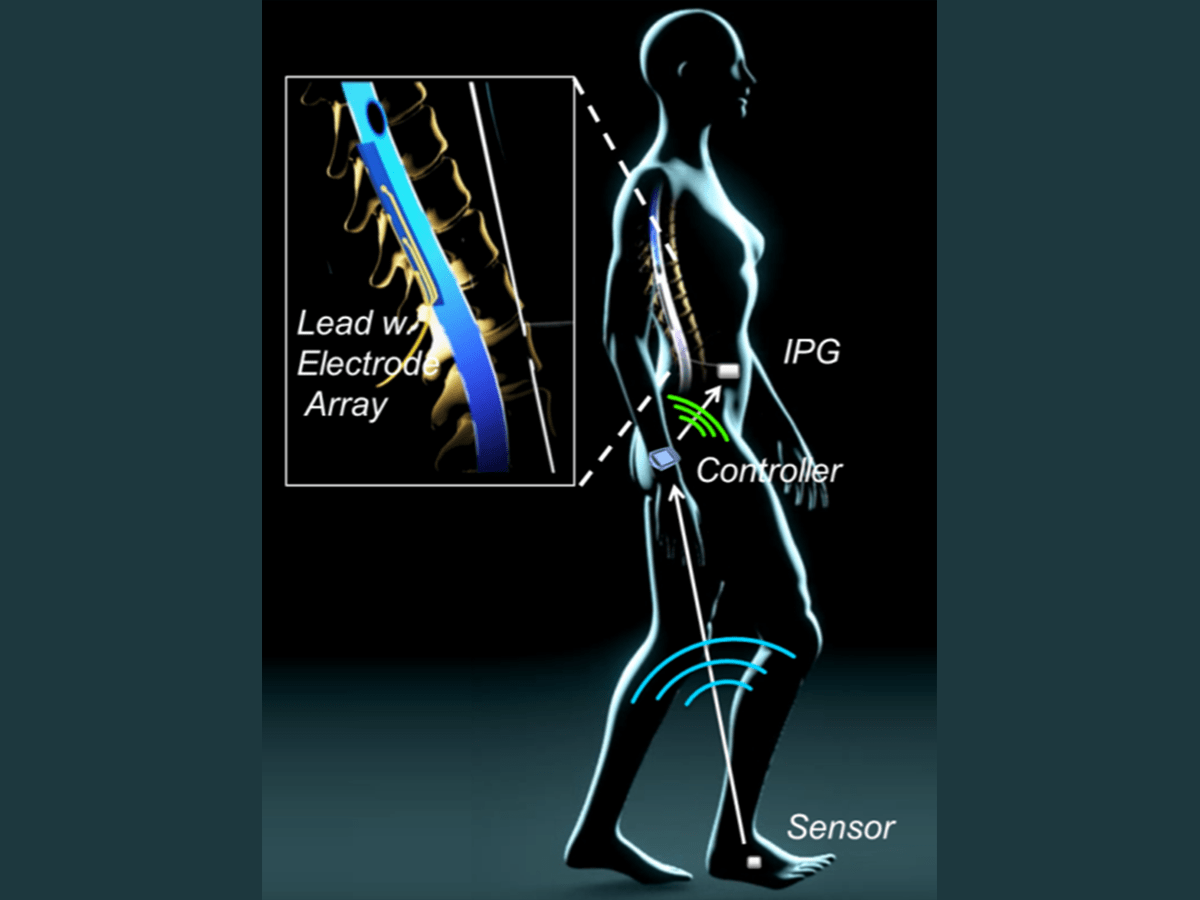
July 7, 2020 NewsThe Food and Drug Administration has granted Gtx Medical BV a breakthrough device designation for its implantable Go-2 Targeted Epidural Spinal Stimulation (TESS) system.
The device enabled patients who had sustained traumatic spinal cord injuries to regain voluntary control over previously paralyzed muscles and walk again four or more years after paralysis, according to a study previously published in Nature.
The company, based in Eindhoven, Netherlands, and Lausanne, Switzerland, plans to start the first clinical trial for TESS in 2021.








Feel free to contribute!